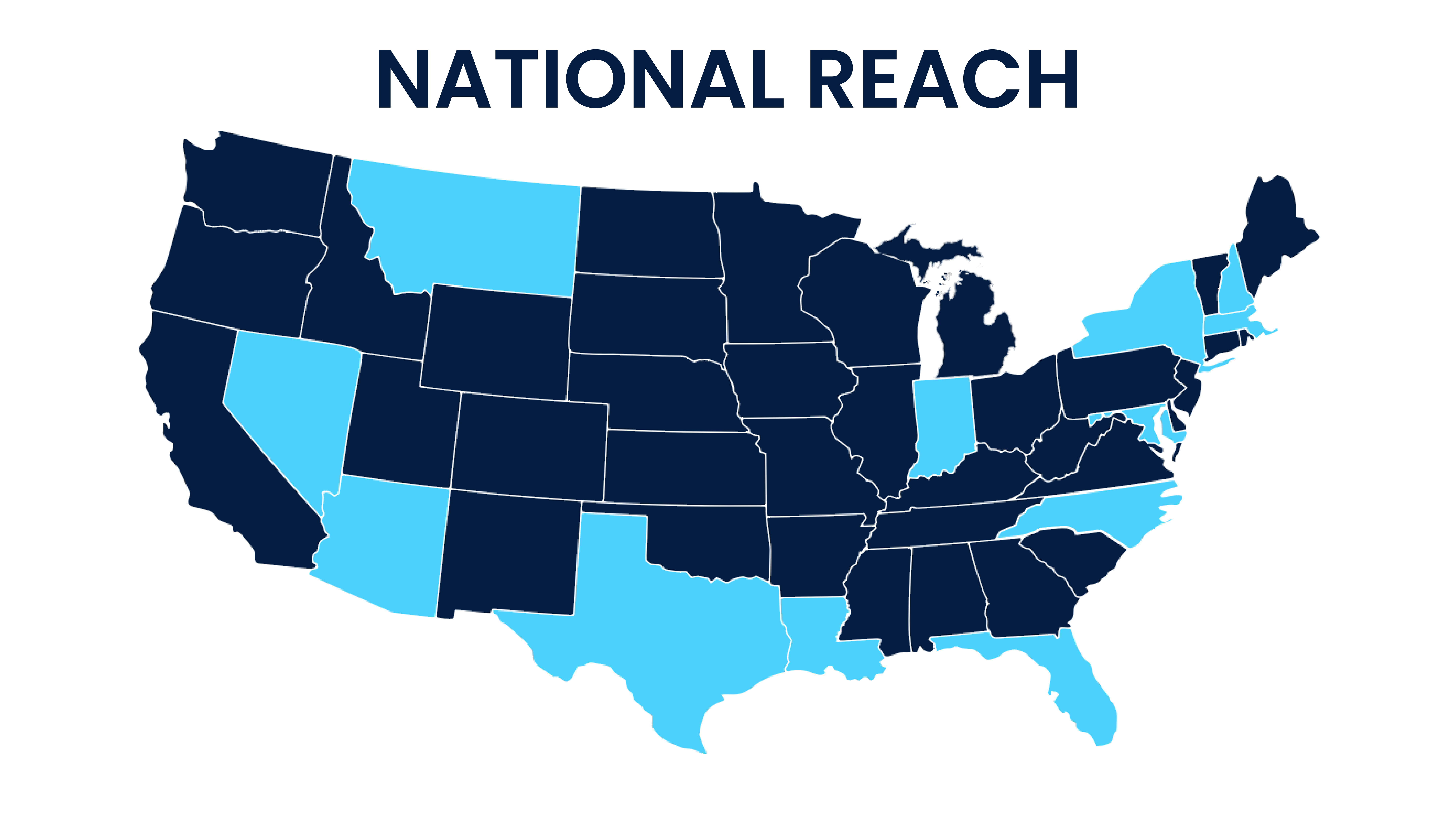
AWARE for All
AWARE for All is an educational program that partners with organizations to bring accessible clinical research information to the public. Each year, our team travels across the U.S. to partner with 15+ events.
View our upcoming events below!